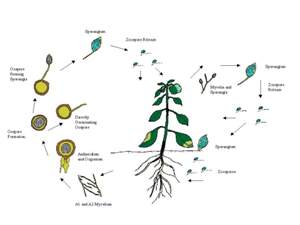
Introduction
Phytophthora capsici Leonian (1922)
Phytophthora capsici was first reported by Leonian on chili pepper (Capsicum frutescens) in
Cultural Characteristics
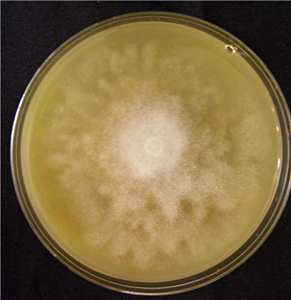
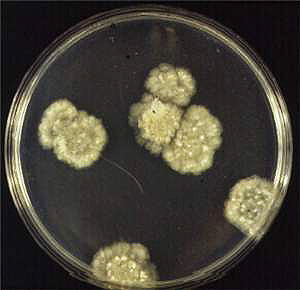
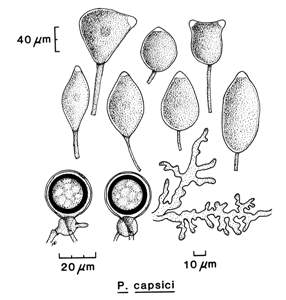
Cultures are radiate and fluffy (Figs. 4 and 5). The minimum temperature for growth is 10�C, the optimum temperature for growth is 28�C, and the maximum temperature for growth is higher than 35�C.
Reproductive Structures
Asexual Structures
Sporangiophores:
Sporangiophores are narrow, widening slightly at the base of the sporangium
and branching irregularly. Simple sympodium forms in water or may be umbellate in tropical isolates.
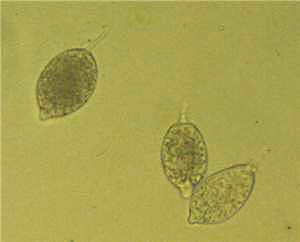
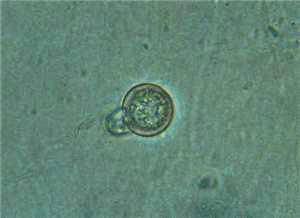
Sporangia:
Sporangia are caducous and often irregular, nearly spherical, broadly ovoid,
and obturbinate or elongated (Fig. 6). Sporangia are papillate and large. Most tropical isolates (CapB) produce ellipsoid-lanceolate
sporangia. They are
17.4�38.7
� 32.8�65.8 µm (Fig. 6). They are often distorted and have more than one apex
and are papillate. The pedicel is long and
is 10 µm or more in length
and up to 100 µm in some isolates.
Chlamydospores:
Chlamydospores are rare or absent in most CapA isolates from pepper but abundant in tropical isolates from black pepper and cacao (CapB isolates). When present they
are 28�29 µm in diameter with a wall thickness of 2.4�2.7 µm.
Hyphae:
Hyphae are fairly coarse and 5�7 µm wide. Hyphal swellings are occasionally produced by some isolates in aqueous cultures.
Sexual Structures
P. capsici is heterothallic. There are two mating types in P. capsici, A1 and A2. Each mating type produces hormones that are responsible for gametangia differentiation in the opposite mating type.
Antheridia:
Antheridia are amphigynous and 15
� 17 µm.
Oogonia:
Oogonia
appear frequently in single cultures of some isolates, but in others, they are only on pairing A1 and A2 mating types. The wall of the oogonia is yellowish brown. Oogonia are spherical and can vary in size from 23 to 50 µm. Oospores are plerotic.
Oospores:
Oospores nearly fill
the oogonium and are thin walled (2�6 µm) (Fig. 7).
The mean oospore diameter of pepper isolates is 23.7�34.9 µm (Ristaino, 1990). Size of the oospore varies with different hosts.
Host Range and Distribution
P. capsici infects 51 genera in 28 plant families. P. capsici causes a devastating disease on bell pepper (Capsicum annuum) and cucurbit (Cucumis sativus) crops in the
|
Host |
Common Name |
Disease |
Geographical Distribution |
|
Capsicum annuum |
|
Blight of leaves, fruits, and stems |
Worldwide |
|
Capsicum frutescens |
Chili
pepper |
Blight of leaves, fruits, and stems |
Worldwide |
|
Cucurbitaceae |
Cucumber, squash |
Blight of fruits and stems |
Worldwide |
|
Gossypium spp |
Cotton |
Blight of leaves, bolls, and stems |
|
|
Lycopersicon esculentum |
Tomato |
Blight of leaves, fruits, and stems |
Worldwide |
|
Macadamia integrifolia |
Macadamia nut |
Blight of leaves, fruits, nut, and hull |
|
|
Piper nigrum |
Black pepper |
Blight of leaves, fruits, and stems |
|
|
Theobroma cacao |
|
Pod rot |
Brazil, Mexico, Cameroon, Costa Rica |
|
Sechium edule |
Eggplant |
Blight of leaves, fruits, and stems |
Costa Rica |
|
Spondia purpurea |
|
Blight of leaves, fruits, and stems |
Costa Rica |
Symptoms
P. capsici causes stem and fruit rot of Capsicum
spp. and soft rot of tomato and cucurbit fruits (Figs. 8�13). P. capsici causes a root and crown rot, as well as an aerial blight of leaves, fruit, and stems, on bell pepper (Capsicum annuum), tomato, cucumber, watermelon, squash, and pumpkin (Ristaino and Johnston, 1999). Oospores are believed to provide the initial source of inoculum in the field, and the disease is polycyclic within seasons. On the pepper fruit, the disease appears first as a small, dull green, water-soaked spot that quickly spreads, covering up to more than one-fourth of the fruit. The infected fruits can
develop lesions that are typically covered with white sporangia. Tissue may become dry and sunken and turn a straw color after exposure to the disease. On the leaves, circular, grayish brown, water-soaked lesions are common.
Fruit
Rot and Root Rot of Cucurbits:
Disease in tomatoes and
cucurbits can manifest in one of two ways. Small, reddish brown lesions or discolorations can
appear on the fruit. These areas grow and become dark green, eventually becoming brownish with irregular margins.
Large, brown, water-soaked lesions can also appear with irregular margins, eventually creating well-defined margins on fruit.
Blight of Macadamia:
P. capsici can infect the flower and the nut of macadamia. P. capsici initially causes a dark brown to black discoloration on the nut.
Sometimes it can penetrate under the husk of the nut and kill the kernel.
Buckeye
Rot, Root Rot, and Crown Rot of Tomato:
Symptoms include irregularly shaped, water-soaked lesions on the fruit with a bull's-eye pattern. As
the diseased area expands, the center becomes dark brown to black and newly infected areas turn light brown. Disease can also be detected by the rapid wilt and complete collapse of younger plants; cankers develop along the roots of older plants, followed by the wilting and necrosis of leaves.
Diagnostics:
Traditionally, P. capsici has been recovered by isolation of the pathogen from infected tissue and plating onto a semiselective medium. The pathogen can also be isolated from the soil following soil dilution plating in dilute water agar onto a semiselective medium such as Masago's medium. Also, a leaf disk baiting assay, in which circular disks of leaves are floated on saturated soil, provides a high level of pathogen detection. Leaf disks can also be plated onto semiselective media for detection. A polymerase chain reaction (PCR) assay has also been developed for detection of the pathogen (Ristaino and Johnston, 1999).
References
Aragaki, M., and Uchida, J. Y. 2001. Morphological distinctions between Phytophthora capsici and P. tropicalis sp. nov. Mycologia 93:137-145.
Erwin, D. C., and Ribeiro, O. K. 1996.
Phytophthora Diseases Worldwide. American Phytopathological Society,
Leonian, L. H. 1922. Stem and fruit blight of peppers caused by Phytophthora capsici sp. nov. Phytopathology 12:401-408.
Mchau, G. R. A., and Coffey, M. D. 1995. Evidence for the existence of two distinct subpopulations of P. capsici and a redescription of the species. Mycol. Res. 99:89-102.
Ristaino, J. B. 1990. Intraspecific variation among isolates of Phytophthora capsici from pepper and cucurbit fields in North Carolina. Phytopathology 80:1253-1259.
Ristaino, J. B., and Johnston, S. B. 1999. Ecologically based approaches to management of Phytophthora blight on bell pepper. Plant Dis. 83:1080-1089.
Stamps, J. 1984. Phytophthora capsici. CMI Descr. Pathog. Fungi Bact. 836:1.
Stamps, D. J., Newhook, F. J., Waterhouse, G. M., and Hall, G. S. 1990. Revised
tabular key to the species of Phytophthora de Bary.
Mycol. Pap. 162.
CAB International,
Wallingford, United Kingdom;
Commonwealth Mycological Institute, Kew
Tsao, P. H. 1991. The identities, nomenclature and taxonomy of Phytophthora isolates from black pepper. Pages 185-211 in: Diseases of Black Pepper. Proc. Int. Pepper Community Workshop on Black Pepper Diseases. Y. Sarma and T. Premkumpr, eds. National Research Centre for Spices, Calicut, Kerala, India.
Tsao, P. H., and Alizadeh, A. 1988. Recent advances in the taxonomy and nomenclature of the so-called "Phytophthora palmivora" MF-4 occurring on cocoa and other tropical crops. Pages 441-445 in: Proc. Int. Cocoa Res. Conf., 10th. Cocoa Producers' Alliance, Lagos, Nigeria.
Tucker, C. M. 1933. The distribution of the genus Phytophthora de Bary. Univ. Mo. Agric. Exp. Stn. Res. Bull. 184.
.jpg)